Chemical Properties of Stomach Acid
|
GCSE CHEMISTRY - Covalent Bonding in a Hydrogen Chloride Molecule - What is the Structure of a Hydrogen Chloride Molecule? - GCSE SCIENCE.. (n.d.). GCSE SCIENCE - The Best Revision for CHEMISTRY and PHYSICS.. Retrieved May 24, 2013, from http://www.gcsescience.com/a29-covalent-bond-hydrogen-chloride-gas-molecule.htm
|
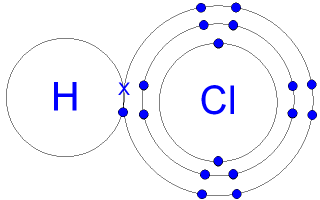
Stomach acid, commonly known as hydrochloric acid, is namely an acid with a pH level ranging from 1.5 to 3.5. Because of the low pH level, stomach acid is highly corrosive when in contact with flesh with no mucus lining. This is why the burning sensation is felt when a victim experiences acid reflux. The acid is burning through the unprotected esophagus. The chemical formula of stomach acid (hydrochloric acid) is HCl. To the left, you can see the covalent bonding of the acid. The hydrogen and chlorine atoms are sharing 2 valence electrons, forming a single bond. Both hydrogen and chlorine have full valence shells, showing it is a stable compound.
|